Research Progress
A single-atom catalyst supported on monolayers for nitrogen fixation
Post: 2017-11-18 22:56 View:2465
| Nitrogen fixation is a process that converts nitrogen in the Earth’s atmosphere into ammonia (NH3). Nitrogen compounds are needed for the biosynthesis of basic building blocks of plants, animals and other life forms, thus nitrogen fixation is essential and crucial to sustain all forms of life. In nature, nitrogen fixation is done by the enzyme nitrogenase, and in industry, the world’s ammonia-based fertilizer is predominantly manufactured by the industrial Haber-Bosch process using H2 and N2. | |||||||||||||
| The main challenge of this process for NH3 synthesis is that it requires extremely high pressures (150-250 bar) and high temperatures (400-500°C) with large amounts of H2 produced by steam reforming of fossil fuels with a large concomitant emission of CO2. | |||||||||||||
| The electrochemical N2 reduction is rather promising for N2 fixation because it can occur at ambient temperature and pressure, in which the NH3 product can be easily separated from hydrogen feed gas, and the nitrogen reduction reaction (NRR) process can be effectively tuned by changing the operating potential, the electrolyte, the pH, etc, thus greatly improving the production yield of ammonia. | |||||||||||||
| Single-atom catalysts (SACs) have emerged as a new frontier in heterogeneous catalysis, and demonstrated distinguishing performances for various reactions due to their high catalytic activity with a significantly reduced amount of metals used. However, the catalytic performance of SACs for N2 fixation and conversion has been rarely explored. | |||||||||||||
|
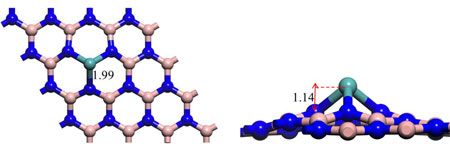
In new work reported in Journal of the American Chemical Society ("Single Mo atom Supported on Defective Boron Nitride Monolayer as an Efficient Electrocatalyst for Nitrogen Fixation: A Computational Study"), Prof. Jingxiang Zhao (College of Chemistry and Chemical Engineering of Harbin Normal University, China) and Prof. Zhongfang Chen (Department of Chemistry, University of Puerto Rico, USA) have proposed a quite promising single-atom-based electrocatalyst for N2reduction to NH3 under ambient conditions.
|
Address: C508 Dingxin Building, Jilin University, 2699 Qianjin Street, Changchun 130012, P. R. China
Copyright © 2024 International Society of Bionic Engineering All Rights Reserved
吉ICP备11002416号-1
Copyright © 2024 International Society of Bionic Engineering All Rights Reserved
吉ICP备11002416号-1